Humans have been increasing the amount of carbon dioxide in air by burning of fossil fuels, by producing cement and by carrying out land clearing and forest combustion. There has been a climb in carbon dioxide concentrations in the atmosphere of about 280 ppm in 1850 to 364 ppm in 1998, mainly due to human activities during and after the industrial revolution, which began in 1850. As a result, it has exceeded the amount sequestered in biomass, the oceans, and other sinks. Carbon dioxide emissions by humansĭue to human activities, the amount of CO 2 released into the atmosphere has been rising extensively during the last 150 years. This chemical behaviour explains why water, which normally has a neutral pH of 7 has an acidic pH of approximately 5.5 when it has been exposed to air. This is how it works:Ĭarbon dioxide dissolves slightly in water to form a weak acid called carbonic acid, H 2CO 3, according to the following reaction:Īfter that, carbonic acid reacts slightly and reversibly in water to form a hydronium cation, H 3O+, and the bicarbonate ion, HCO 3-, according to the following reaction: Influence of carbon dioxide on alkalinityĬarbon dioxide can change the pH of water. The cycle is more clearly present in the northern hemisphere because it has relatively more land mass and terrestrial vegetation. Overall, then, carbon dioxide in the atmosphere decreases during the growing season and increases during the rest of the year.īecause the seasons in the northern and southern hemispheres are opposite, carbon dioxide in the atmosphere is increasing in the north while decreasing in the south, and vice versa. However, both processes occur the entire year. Photosynthesis dominates during the warmer part of the year and respiration dominates during the colder part of the year. 
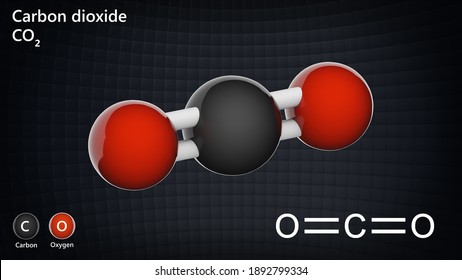
Photosynthesis and respiration play an important role in the carbon cycle and are at equilibrium with one another. This is the respiration process, the reverse of photosynthesis. Plants and animals, in turn, convert the food compounds by combining it with oxygen to release energy for growth and other life activities. The reaction of photosynthesis is as follows: Green plants convert carbon dioxide and water into food compounds, such as glucose, and oxygen. These processes will be briefly explained here. Carbon dioxide plays an important part in vital plant and animal process, such as photosynthesis and respiration. The part carbon dioxide plays in environmental processesĬarbon dioxide is one of the most abundant gasses in the atmosphere. The solid form of carbon dioxide, commonly known as Dry Ice, is used in theatres to create stage fogs and make things like "magic potions" bubble. It prevents oxygen from getting to the fire and as a result, the burning material is deprived of the oxygen it needs to continue burning.Ĭarbon dioxide is also used in a technology called supercritical fluid extraction that is used to decaffeinate coffee. Carbon dioxide can blanket a fire, because of its heaviness. Some fire extinguishers use carbon dioxide because it is denser than air. Carbon dioxide released by baking powder or yeast makes cake batter rise. The most familiar example is its use in soft drinks and beer, to make them fizzy. Humans use carbon dioxide in many different ways. There are several physical and chemical properties, which belong to carbon dioxide. This event is characterised by the CO2 bubbles forming into water. After pressure drops the CO2 gas will try to escape to air. Carbon dioxide is only water-soluble, when pressure is maintained. Liquid carbon dioxide mainly exists when carbon dioxide is dissolved in water. It can only be solid when temperatures are below -78 oC. The linear molecule consists of a carbon atom that is doubly bonded to two oxygen atoms, O=C=O.Īlthough carbon dioxide mainly consists in the gaseous form, it also has a solid and a liquid form. At room temperatures (20-25 oC), carbon dioxide is an odourless, colourless gas, which is faintly acidic and non-flammable.Ĭarbon dioxide is a molecule with the molecular formula CO 2. Joseph Black, a Scottish chemist and physician, first identified carbon dioxide in the 1750s. Carbon dioxide What is carbon dioxide and how is it discovered?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
